top of page

Darby and Cobbs Creek Data
Every four weeks, community science volunteers visit sites throughout the Darby and Cobbs Creek Watershed to take pictures to track changes in erosion and collect data on the following water chemistry parameters:


Water Temperature
Water Temperature is an important water chemistry parameter as it determines the amount of available dissolved oxygen in the waterway - warm water can hold lower concentrations of dissolved oxygen, which means less is available for sensitive stream wildlife.
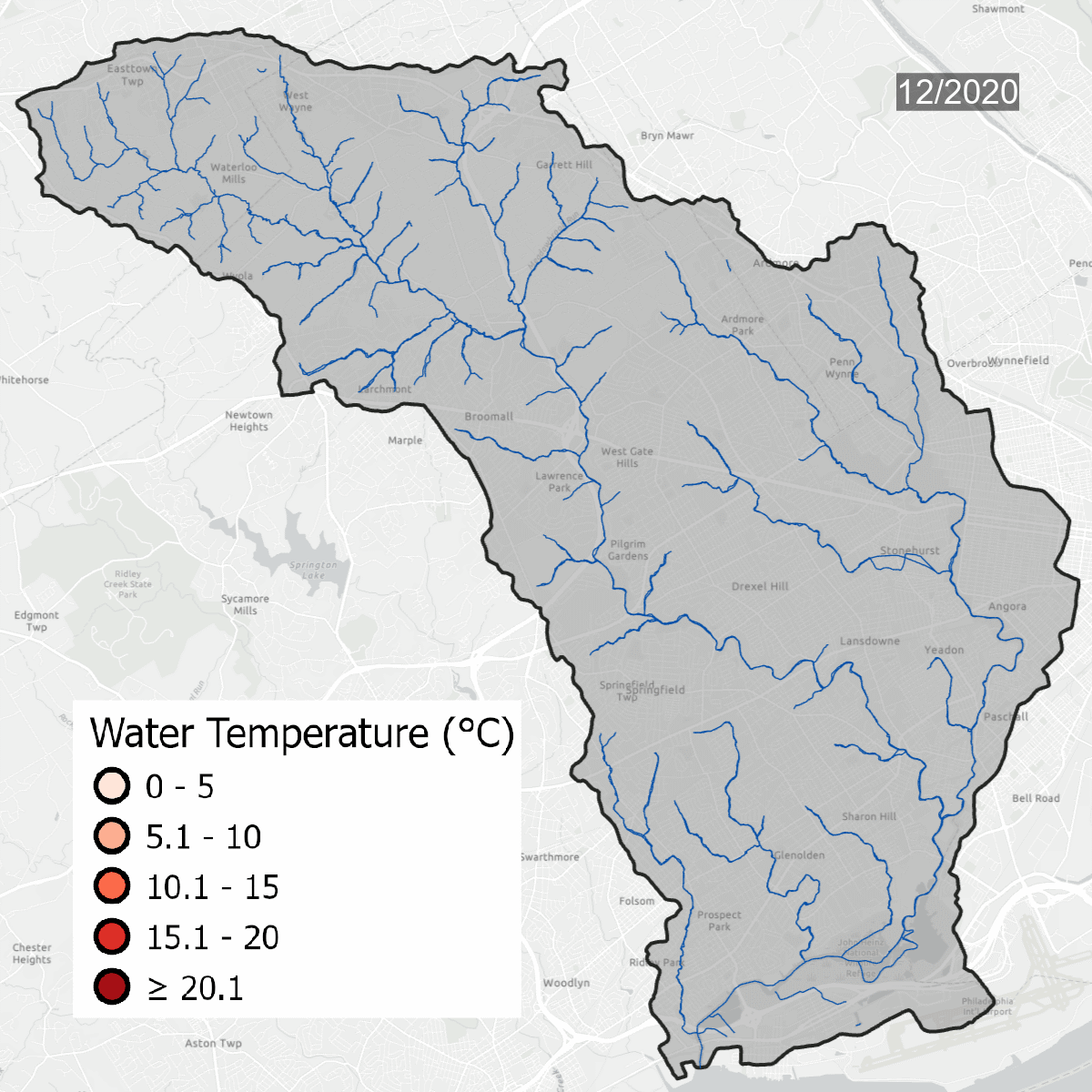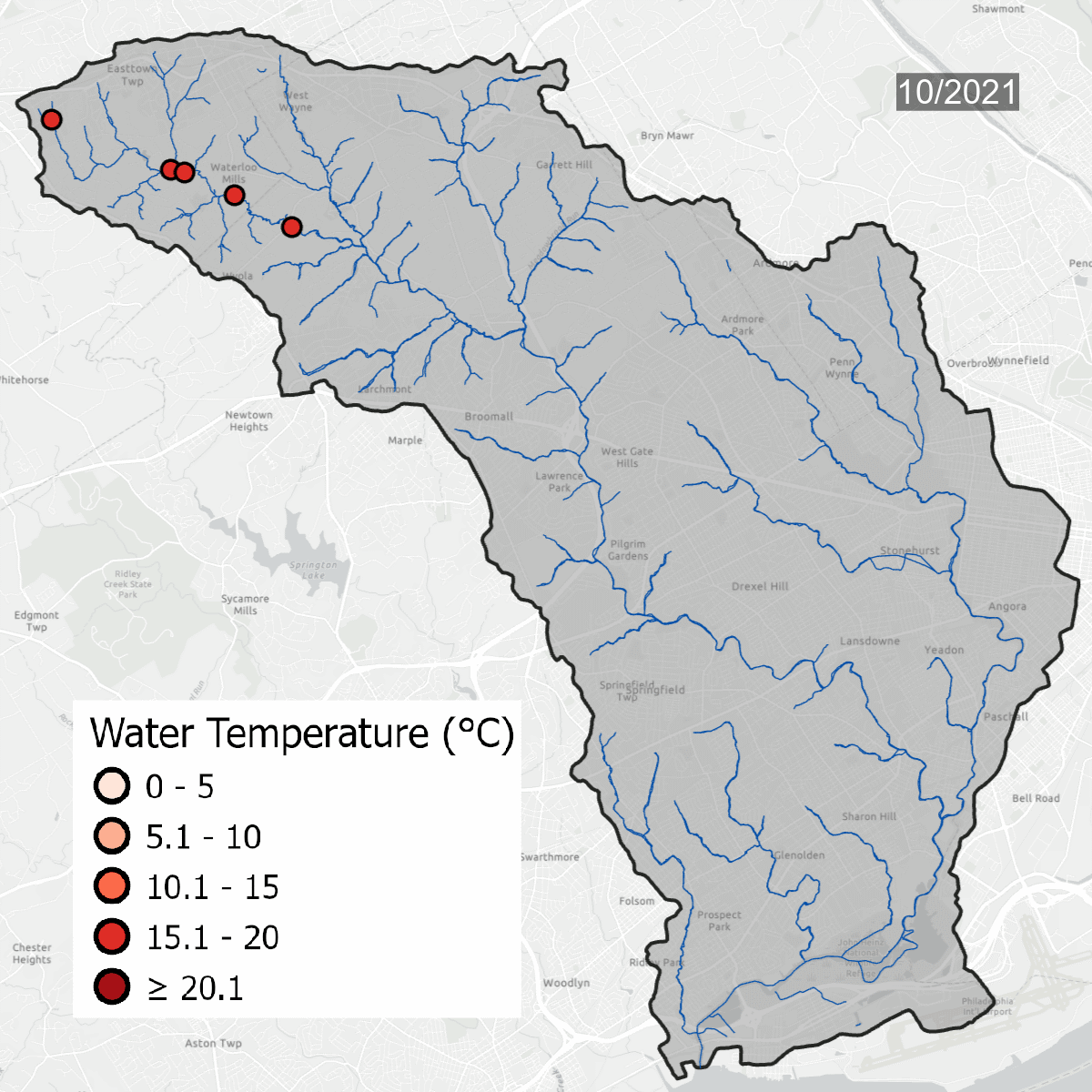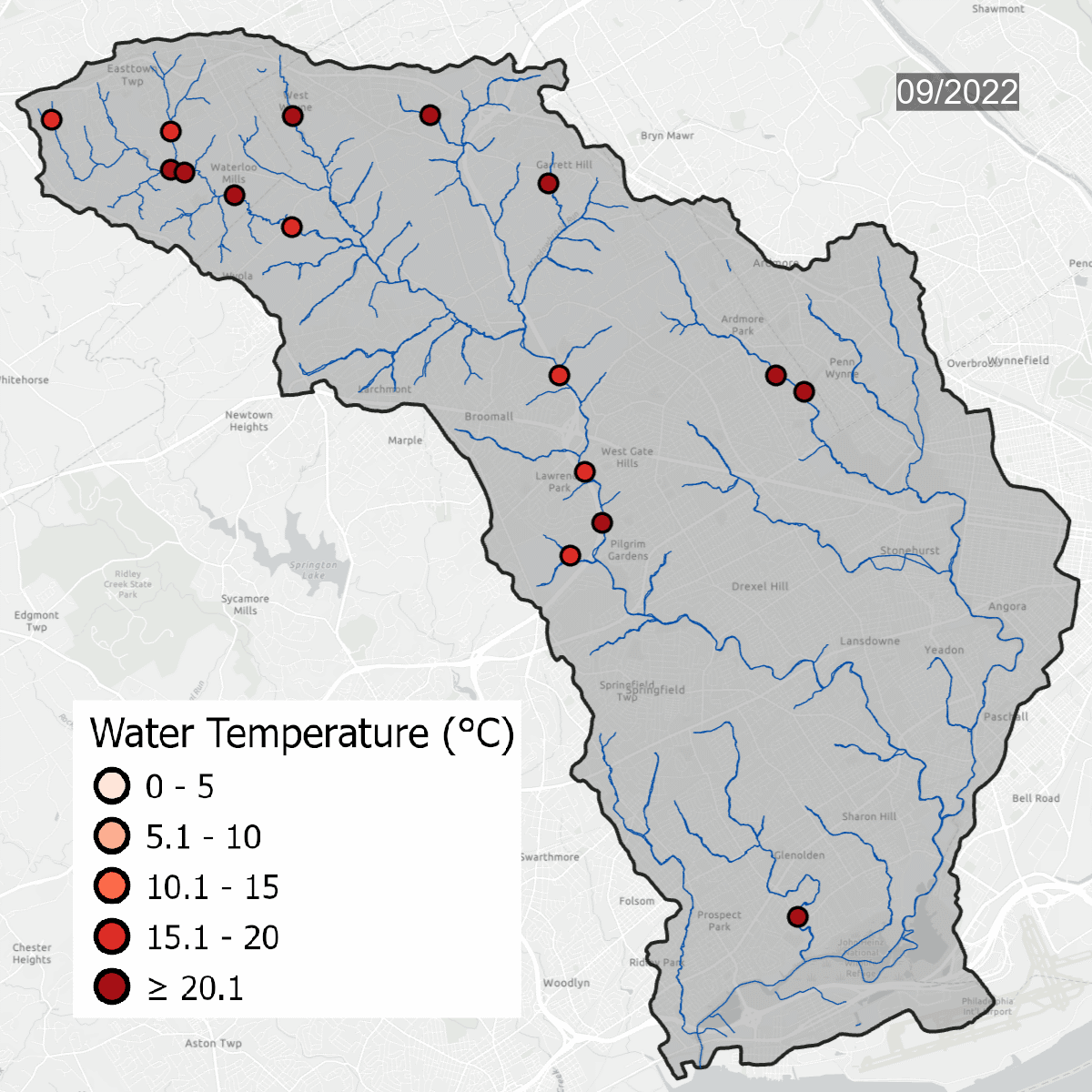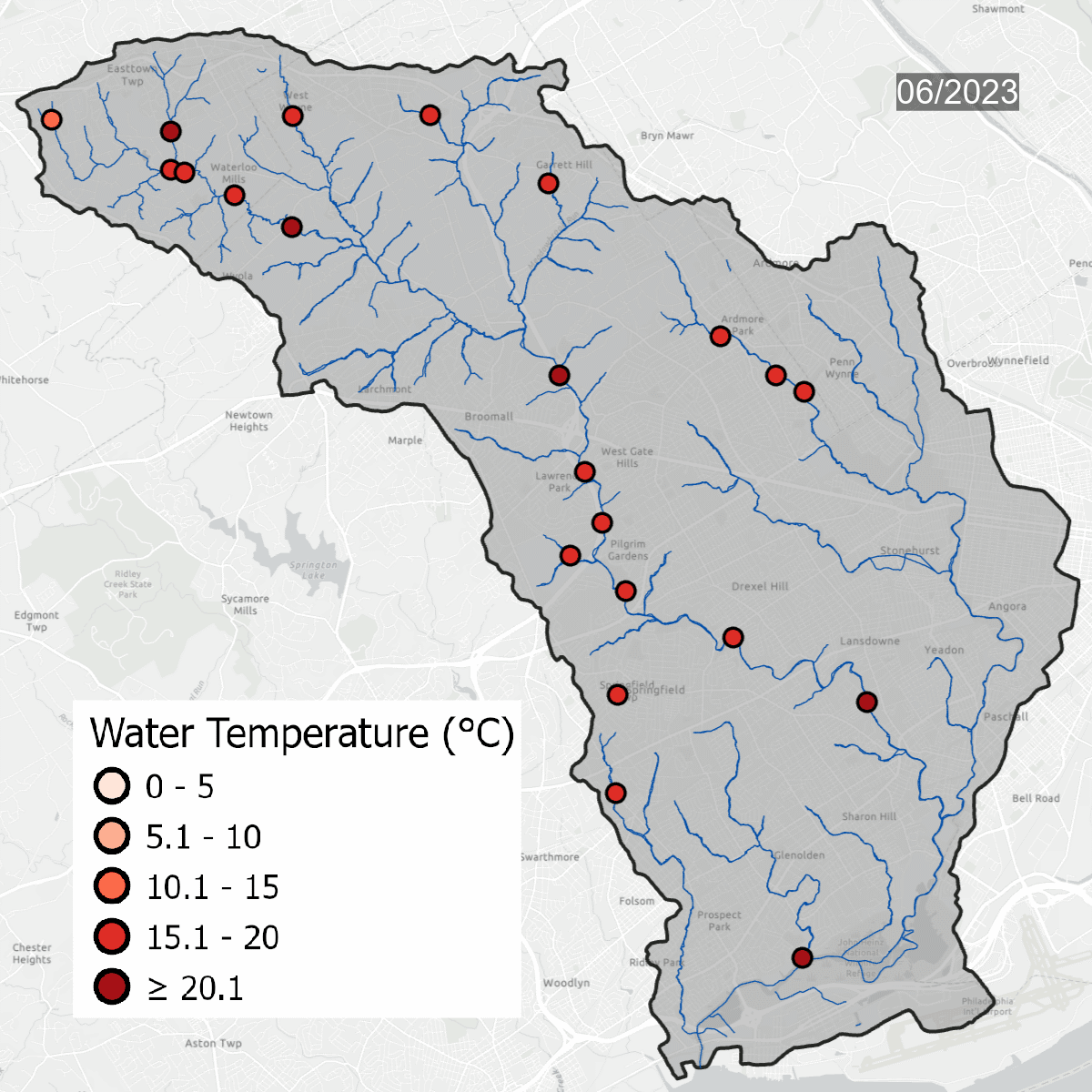
Water temperatures at all sites over time. Warmer water temperatures have darker colors, cooler water has lighter temperatures. Note the seasonal change at sites.

This is the same data that is on the map above, but laid out over the course of several years, which shows seasonal changes - with warmer water in the summer and colder water in the winter. Graph updated with data through September 2023.
Water temperature is one of the major thresholds that the Pennsylvania Department of Environmental Protection uses to determine if a waterway is healthy. Sites need to be regularly below the solid line on the graph to be considered a Cold Water Fishery (CWF). As water becomes warmer, cold tolerant species begin to disappear, changing the dynamic of the waterway. Winter temperatures in the Darby/Cobbs Watershed are almost entirely warmer than the maximum temperature to be considered a Warm Water Fishery (WWF). Winter and early spring are when species are most sensitive to temperature pollution.

Conductivity
Conductivity levels increase as more ions dissolve in the water. In the winter, the application of road salt on driveways, roads, and sidewalks drives higher levels of conductivity in waterways - while salt is not the only driver of higher conductivity, it is the major contributor in the region.
Melting winter snow and ice carries salt into streams, causing rapid increases in conductivity levels throughout the watershed. Rainstorms can cause conductivity levels to fall, as more water separates the ions, reducing the ability of the water to carry an electric current.
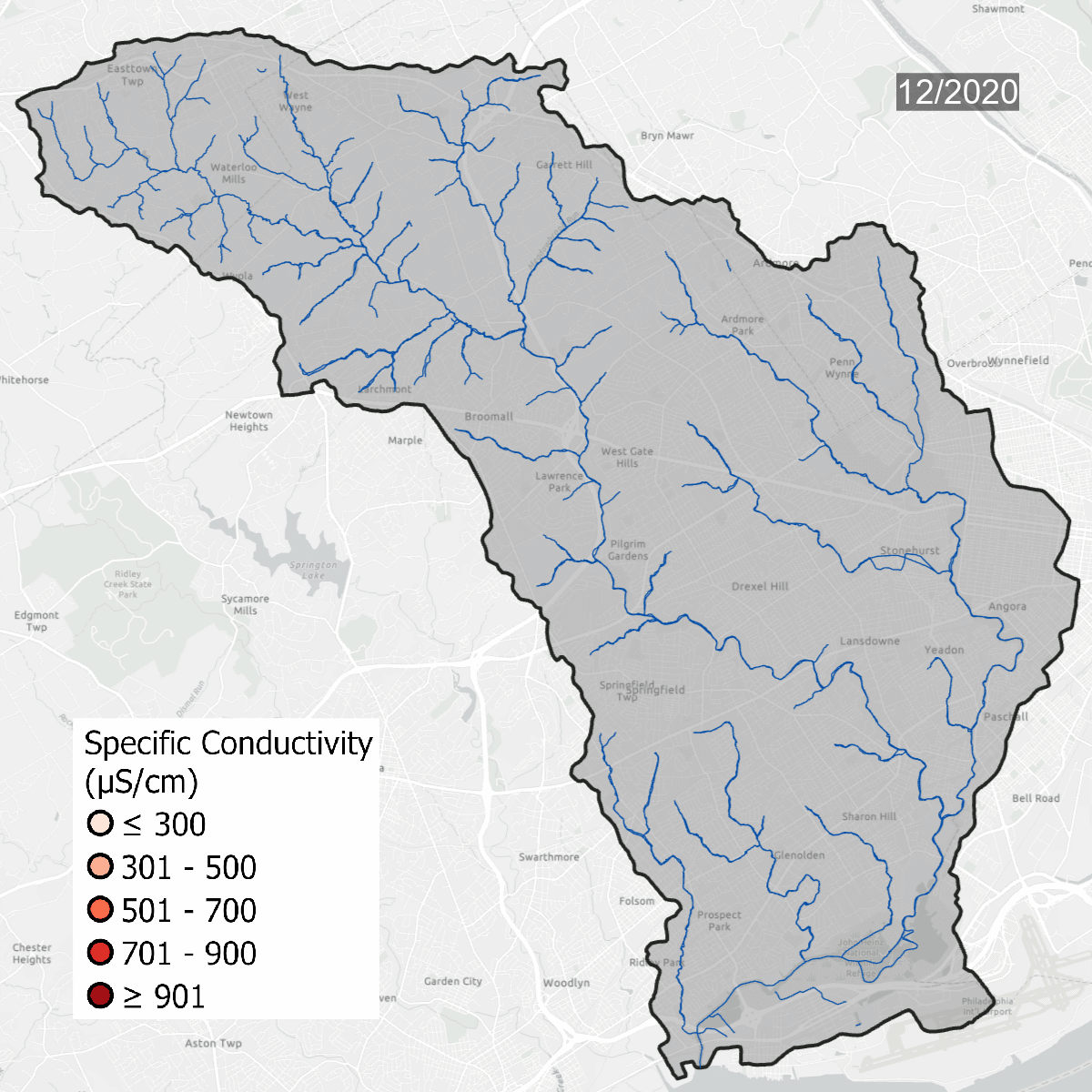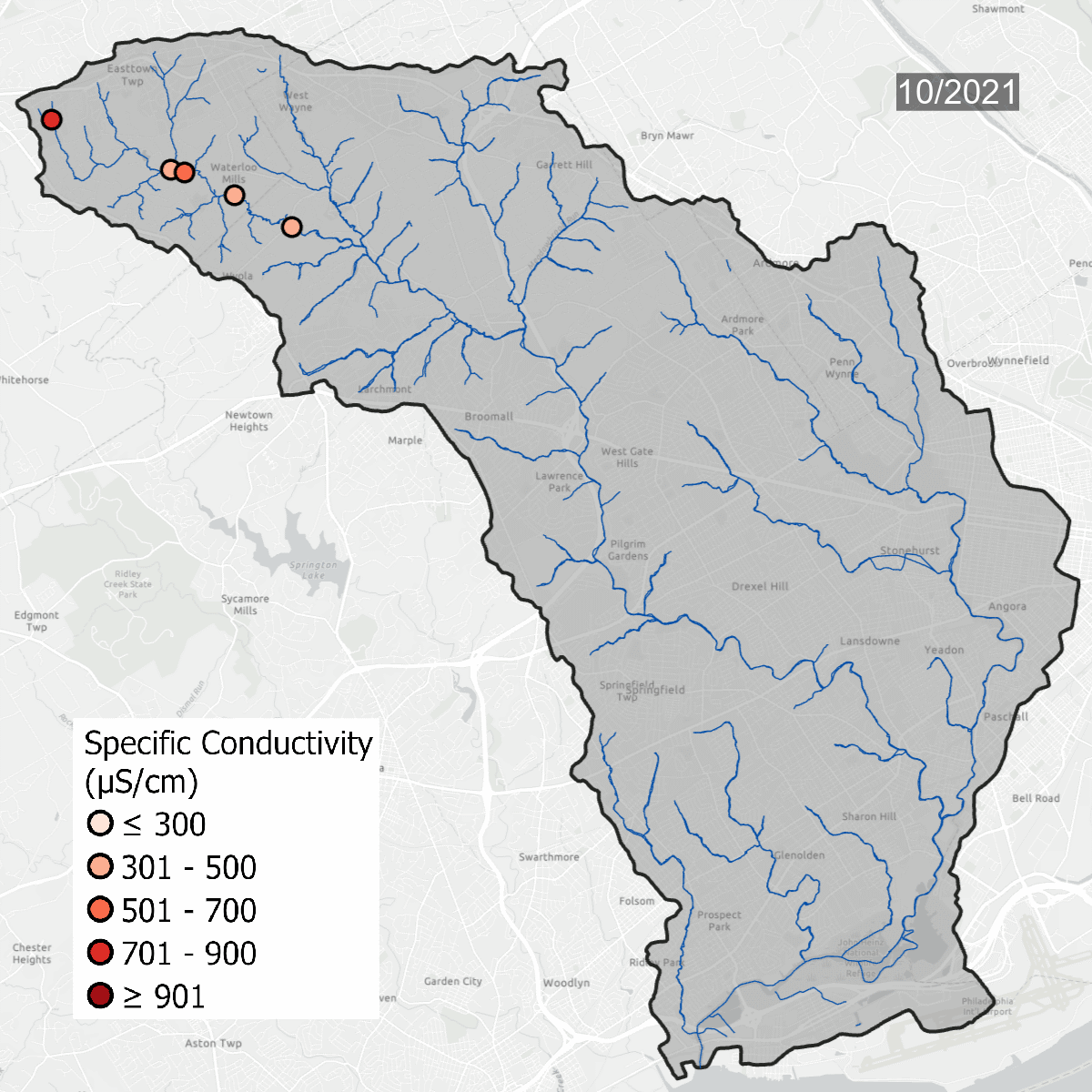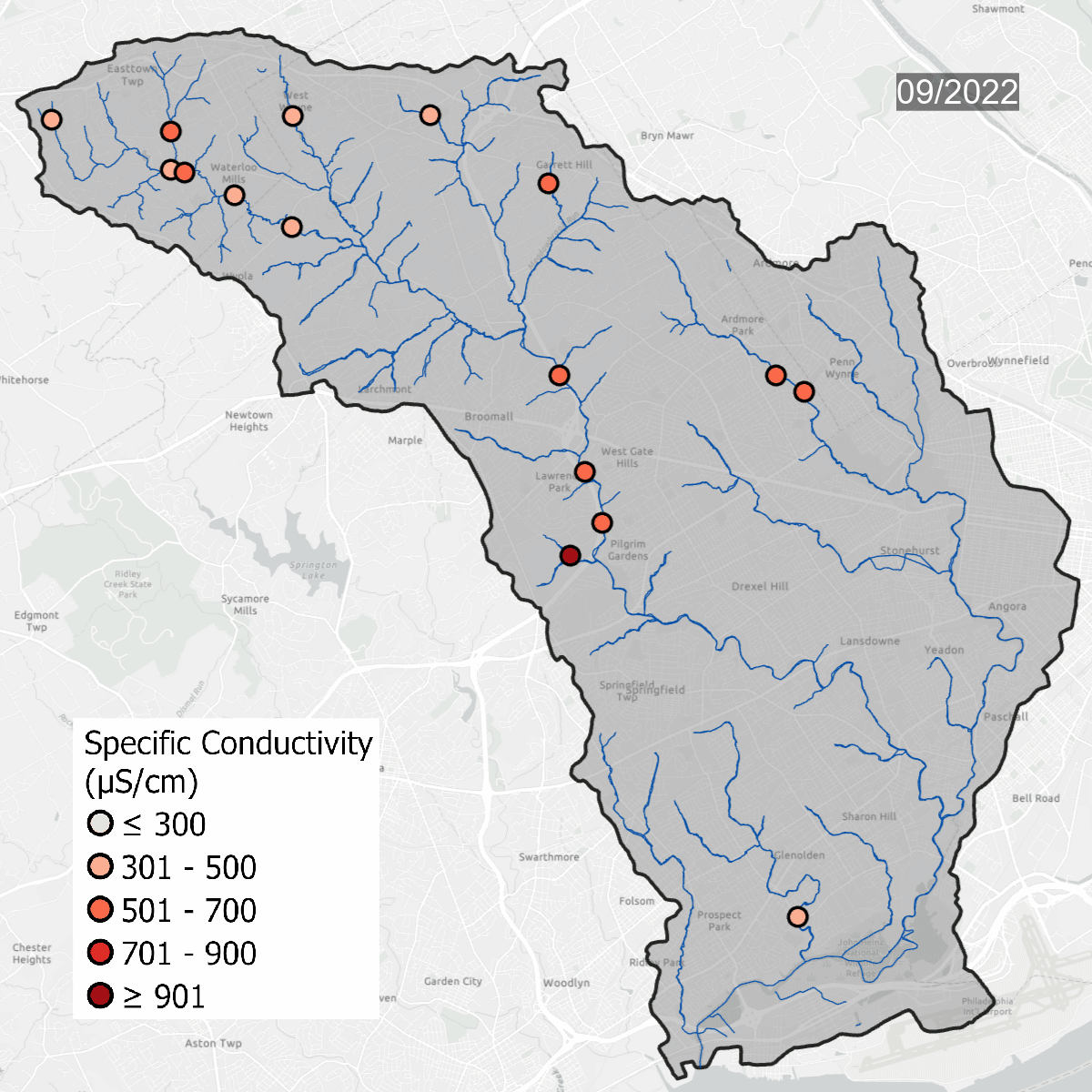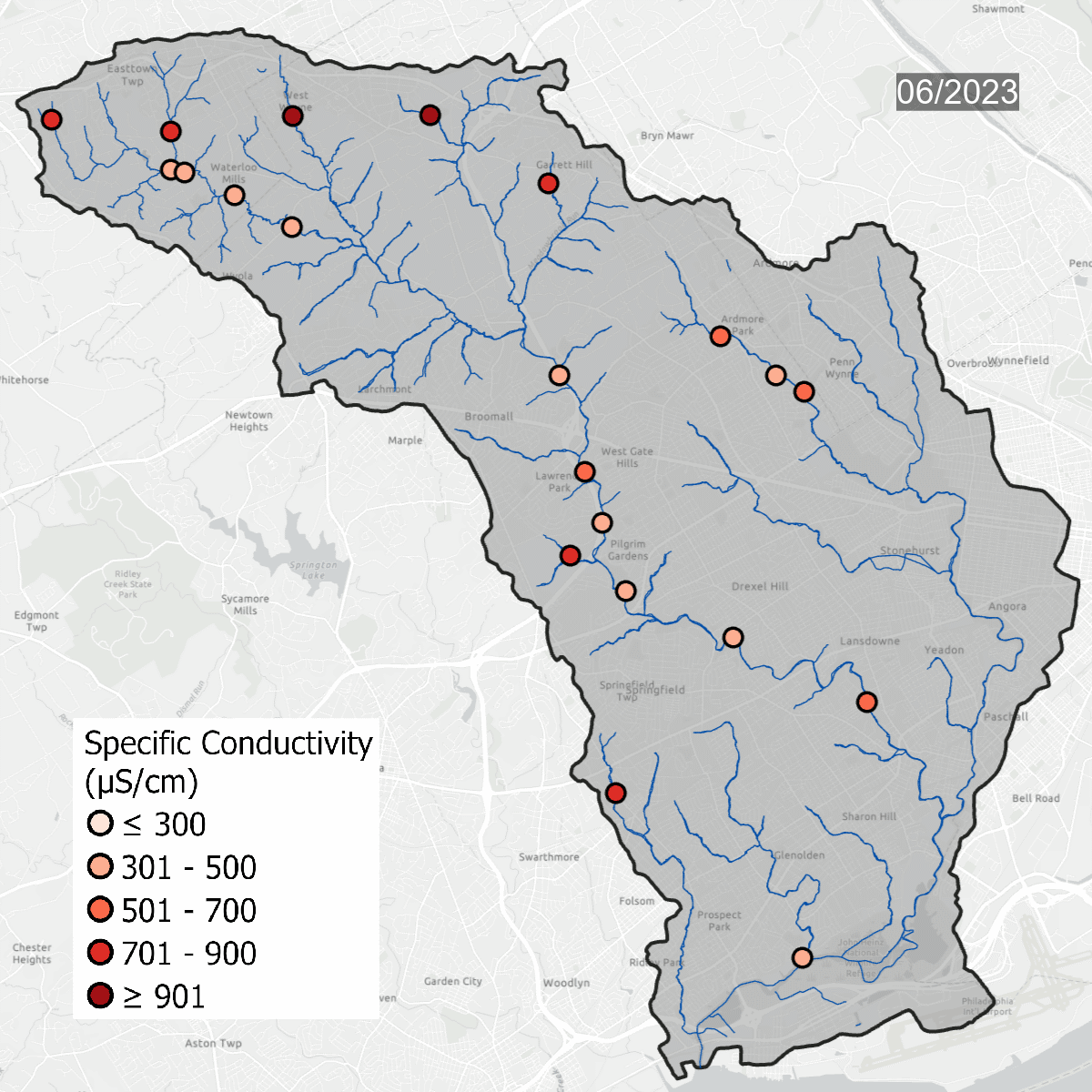

Conductivity levels at all sites over time. All sites show that conductivity levels are far above baseline, which should be between 75-90 uS/cm.
Conductivity is measured using a Hanna Conductivity meter.

This is the same data that is on the map above, but laid out over the course of several years, which shows the range of conductivity levels throughout the course of the project. Graph updated with data through September 2023.
Spikes in conductivity occur in the winter months after snow and ice melt and carry road salt into waterways. Some sites, like DCYM1 in pink, have conductivity levels that are always high due to the build up of salts and metals in the soil.
The more development on the landscape, the higher conductivity will be over time.
The Darby and Cobbs Creek watersheds become highly developed the closer you travel to the Delaware River, but any development upstream causes changes in water chemistry. Changes in the headwaters will impact the lower watershed, which is already highly influenced by a long history of landscape changes and industrial development.

Chloride
Chloride is introduced into the watershed most often through the application of road salts (usually sodium chloride, NaCl). When salt dissolves, it separates into the two elements. Chloride is very easy to test for using Quantab Chloride Strips.

Chloride concentrations over time.
- 50 mg/L is the ecological threshold for impact on wildlife
- 120 mg/L is Canada's long-term exposure limit
-
Long-term or chronic exposure means concentrations should remain below this threshold to protect most aquatic life
- 230 mg/L is the USEPA's long-term exposure limit
-
250 mg/L is when you can taste salt in water
- 640 mg/L is Canada's acute exposure limit
- The threshold that aims to protect most aquatic life from severe transient exposure like heavy salt runoff after a storm event
- 860 mg/L is the USEPA's acute exposure limit

This is the same data as presented in the gif above, but plotted out over time. Chloride levels increase in winter months, but with regular application can build up in the soil and ground water causing high levels to be present year round. The orange line is at 50 mg/L, the level where continuous exposure can impact stream life. The red line marks 230 mg/L, the USEPA's long term exposure limit. The graph updated with data through September 2023.

Chloride plotted against the amount of impervious surface on the landscape in the watershed of each site. As impervious surface increases, so does the amount of chloride in each sample.
Orange sites are part of the Darby Creek Community Science Program. Teal sites are part of the Willistown Conservation Trust's monthly water monitoring.
Chloride levels increase in relation to the amount of surfaces that do not allow for water to pass through. With more slippery surfaces, more road salt is applied in excess, washing into nearby streams and collecting in soils. Salt use is on the rise nation wide, which means more stressful conditions in waterways for sensitive life.

20 million metric tons of salt are applied to roads in the United States of America annually.

pH
pH determines how easily aquatic organisms can use nutrients and indicates how toxic pollutants may be. A pH range between 6 and 9 is ideal for aquatic life to thrive, when the range is above this (more basic) or below (more acidic), nutrients are harder to absorb and pollutants become more toxic.

pH plotted over time. pH is measured using Panphea pH strips, which reads every 0.5 pH units, giving the graph an unusual look compared to the other figures. Stream pH is influenced by in-stream photosynthesis, local geology and human based geology.
bottom of page


